Development of a Combination Therapy for Non-Small Cell Lung Cancer (NSCLC)
Treatment of non-small cell lung cancer patients without targetable oncogenic mutations and without PD-L1 expression remains a major challenge with an unmet medical need for new, rational therapeutic strategies (1).
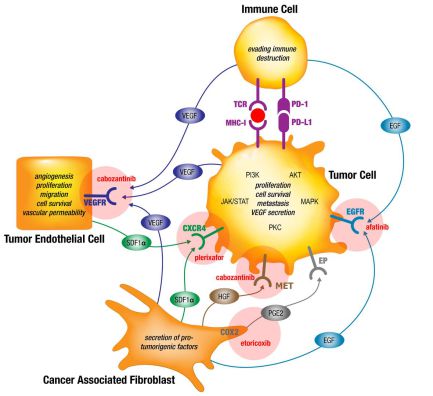
Tumors can be conceived as organs constituted of different cell types with distinct biologic functions, building a Cellular Tumorigenic Network with integrated inter- and intracellular signaling. Next to the neoplastic cells, originating from critical genetic alterations, non-neoplastic cells such as cancer associated fibroblasts, endothelial cells and immune cells comprise a heterogenic tumor microenvironment. Tumor cells are able to reprograms those associated cell types inducing processes such as neoangiogenesis, inhibition of apoptosis, immune suppression and induction of hypoxia and senescence. During the evolving tumor development and as response to targeted therapies, the Cellular Tumorigenic Network is reshaped further leading to heterogeneity and adaptive drug resistance in many cases (2,3,4,5). This is partly based on substitutions for inhibited pathways and on crosstalk of generic intracellularly connected downstream pathways such as the mitogen-activated protein kinase (MAPK), Janus kinase and the signal transducer and activator of transcription protein (JAK/STAT), phosphoinositide 3-kinase (PI3K), protein kinase B (AKT) and protein kinase C (PKC) (6).
The simultaneous and distinct inhibition of signaling within the Cellular Tumorigenic Network and suppression of cellular interdependency by targeting critical paracrine signaling axes is intended to inhibit tumor cell proliferation and thus to overcome drug resistances (7).
Based on this hypothesis we compiled a low-dose targeted drug regimen combining drugs inhibiting tumor-, endothelial-, immune- and cancer associated fibroblast cell interaction by blocking VEGFR and HGFR (cabozantinib), CXCR4 (plerixafor), EGFR (afatinib) and COX2 (etoricoxib). This parallel inhibition of paracrine signaling should effectively inhibit the generic intracellularly connected downstream MAPK, JAK/STAT, PI3K, AKT and PKC pathways.
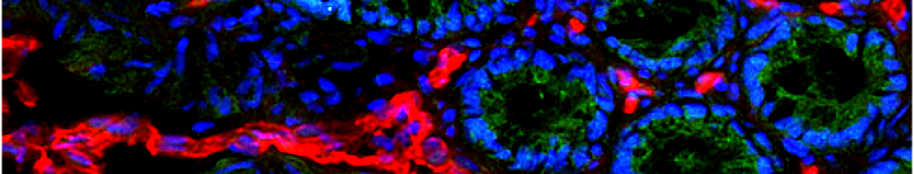
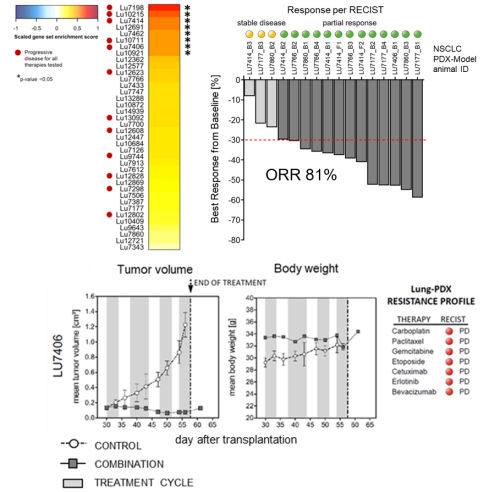
For experimental validation of this hypothesis, five highly resistant patient derived non-small cell lung cancer xenograft models have been selected from a panel of more than thirty available tumors and subjected to treatment with the proposed drug combination regimen. All sixteen patient-derived lung cancers, including highly therapy-resistant adeno- and squamous cell carcinomas without targetable oncogenic mutations, were completely growth suppressed by this drug regimen, leading to an Objective Response Rate of 81% and a Clinical Benefit Rate of 100% with an excellent safety profile. Bioinformatics analysis of gene expression profiles for these lung cancer models provides evidence of a relationship between the expression of the paracrine signaling pathway members and common drug resistances which could be overcome by the new drug combination.
These results strongly encourage the further validation of this cabozantinib, afatinib, plerixafor and etoricoxib combination therapy in preclinical and clinical studies for advanced stage lung cancer patients without current therapeutic options (Gürgen et a., 2022).
References
(1) Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature 553, 446-454 (2018).
(2) Egeblad M, Nakasone ES, Werb Z. Tumors as organs: complex tissues that interface with the entire organism. Dev Cell 18, 884-901 (2010).
(3) Hanahan D. Hallmarks of Cancer: New Dimensions. Cancer Discov 12, 31-46 (2022).
(4) Ohlund D, Elyada E, Tuveson D. Fibroblast heterogeneity in the cancer wound. J Exp Med 211, 1503-1523 (2014).
(5) Hahn WC, Bader JS, Braun TP, et al. An expanded universe of cancer targets. Cell 184, 1142-1155 (2021).
(6) Sever R, Brugge JS. Signal transduction in cancer. Cold Spring Harb Perspect Med 5, (2015).
(7) Langhammer S, Scheerer J. Breaking the crosstalk of the cellular tumorigenic network: Hypothesis for addressing resistances to targeted therapies in advanced NSCLC. Oncotarget 8, 43555-43570 (2017).
Drug Discovery - Oncology
Expanding cancer treatment options - Identification and validation of new tumor-specific and highly functional receptor targets (Langhammer et al., Target Research Group, Bayer Schering Pharma AG, 2007).
Fig. 2
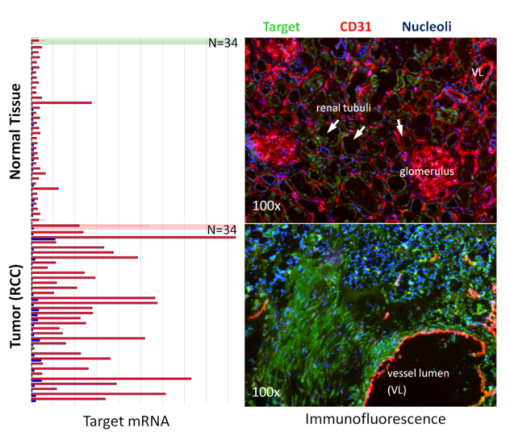
Array Northern analysis of target mRNA and immunofluorescence of target protein in patient samples. Newly identified oncology target is exclusively upregulated in renal cell carcinomas (RCC) of 34 patients analyzed.
Translational Medicine - Virology
Answer to bioterrorist threats - Identification of new therapeutic options for treatment of poxvirus infections by applying translational medicine concepts (Langhammer et al., Antiviral Research, 2011).
Fig. 3
In order to identify clinical relevant targets for the treatment of specific viral infections we established a standardized drug discovery process to identify, test and validate compounds and approved drugs for antiviral treatment.
Fig. 4
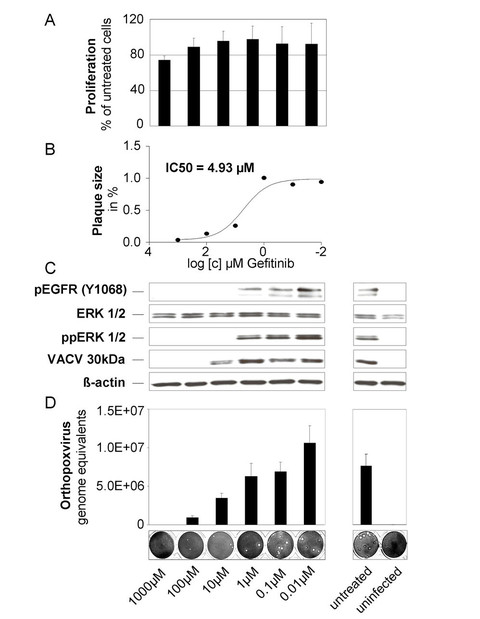
Poxvirus induced EGFR signaling suppressed by the anti-tumor drug Gefitinib is dose dependent and non-toxic at millimolar concentrations. Results from plaque reduction tests of Hep2 cells infected with VACV (12.5 pfu/well) treated with a serial dilution of Gefitinib (1000µm – 0.01µm) and analyzed for (A) proliferation as indicator of cytotoxicity, (B) IC50 of plaque size inhibition, (C) dose dependent inhibition of EGFR-ERK1/2 signaling and VACV proteins by Western blot analysis (pEGFR detection was obtained from a different experiment with identical settings) and (D) dose dependent inhibition of orthopoxvirus genome replication by real-time PCR. Untreated VACV infected cells and uninfected cells are shown as controls in the right panel.
Vaccine Development
Vaccine Development against Retroviruses - First time ever showing successful vaccination by immunization with the transmembrane envelope protein of a retrovirus (Langhammer et al., Antiviral Research, 2011).
Fig. 5
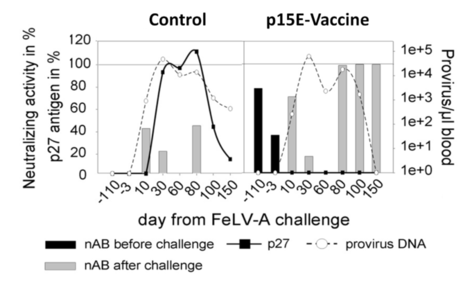
Feline Leukemia Virus (FeLV) antigen p27, provirus DNA and neutralizing activity in a non-immunized (Control) and in a p15E immunized cat (p15E-Vaccine) before and after challenge with the FeLV-A retrovirus. p27 antigen values are expressed as percentage (black line, scale indicated at left y-axis). FeLV neutralizing activity before/after virus challenge is presented in black/grey bars (scale indicated at the left y-axis). FeLV-A provirus integration is shown in copies/µl peripheral blood (dotted line, scale indicated at the right y-axis).
Vaccine Development against HIV-1 - Neutralization capacity of rat antisera induced by immunization with exploratory vaccine candidates (Langhammer et al., Robert Koch-Institute, 2006).
PDF-Dokument [658.5 KB]
Fig. 6
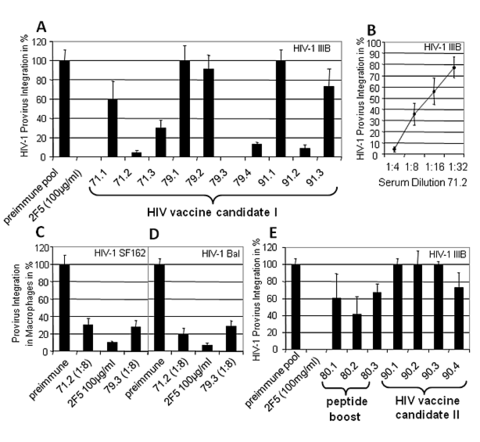
HIV-1 infection was measured as provirus integration by quantitative real time PCR. The mAb 2F5 was used as positive control at a concen-tration of 100µg/ml. A pool of preimmune sera was used as negative control. Provirus integration in the presence of the preimmune serum was taken as 100%. A, Sera obtained from immunizations with efficient HIV vaccine candidate I: Neutralization assay using HIV-1IIIB and C8166 cells, all sera were added at concentration 1:4. B, Neutralization assay using HIV-1IIIB and C8166 cells, serum 1.2 was added at different dilutions. C, D, Neutralization assay using the primary HIV-1 isolates SF162 (C) and Bal (D) and freshly isolated macrophages. Rat immune sera 1.2 and 1.6 were used at a dilution 1:8. E, Sera obtained from immunizations with inefficient HIV vaccine candidate II and from peptide boosts: Neutralization assay using HIV-1IIIB and C8166 cells, rat sera of animal groups 2 and 3 which differed in the boost immunization were added at a concentration 1:4.